About our research
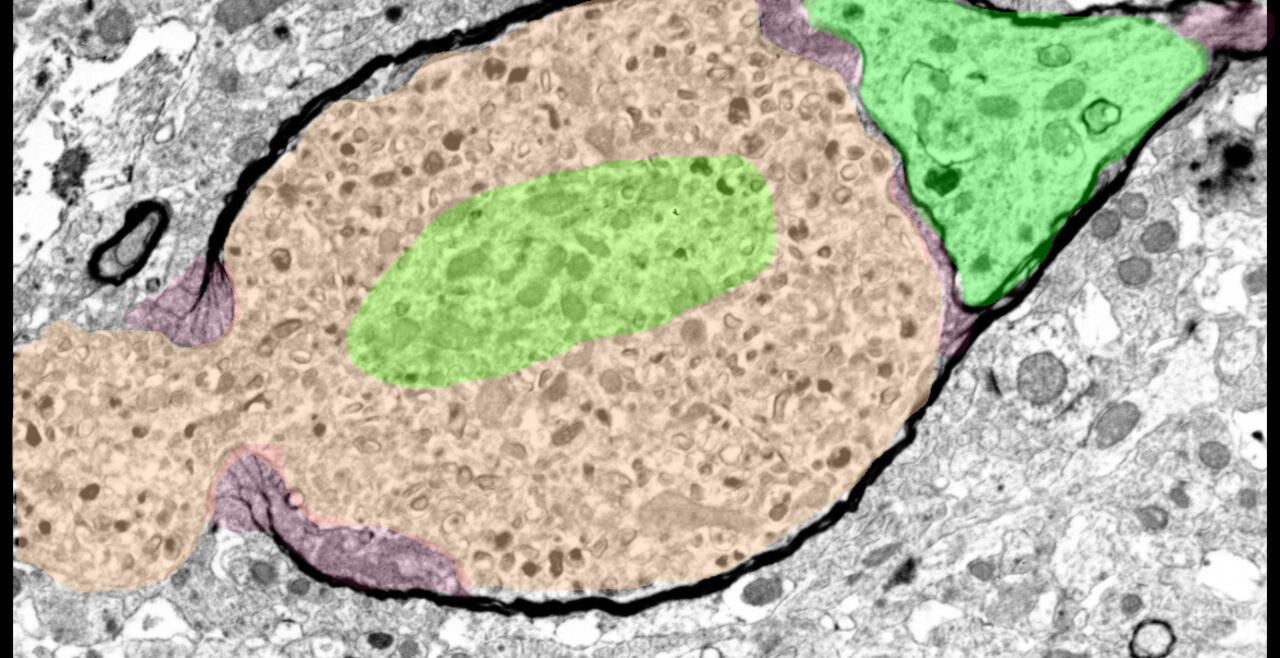
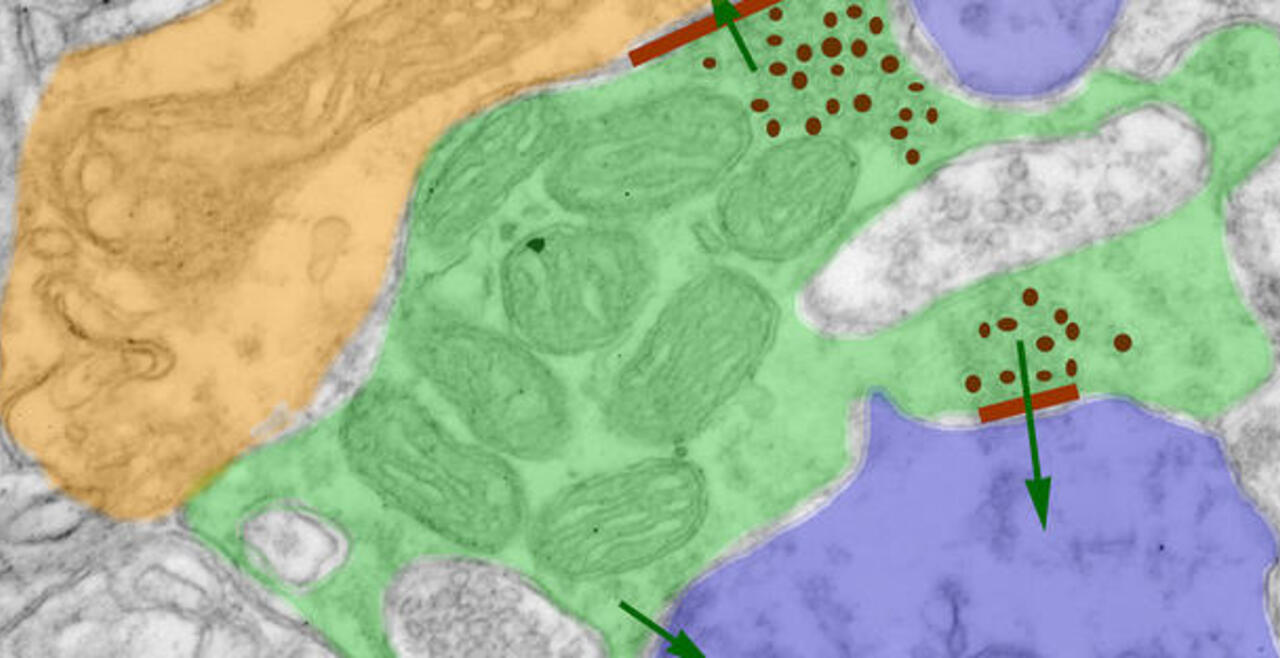
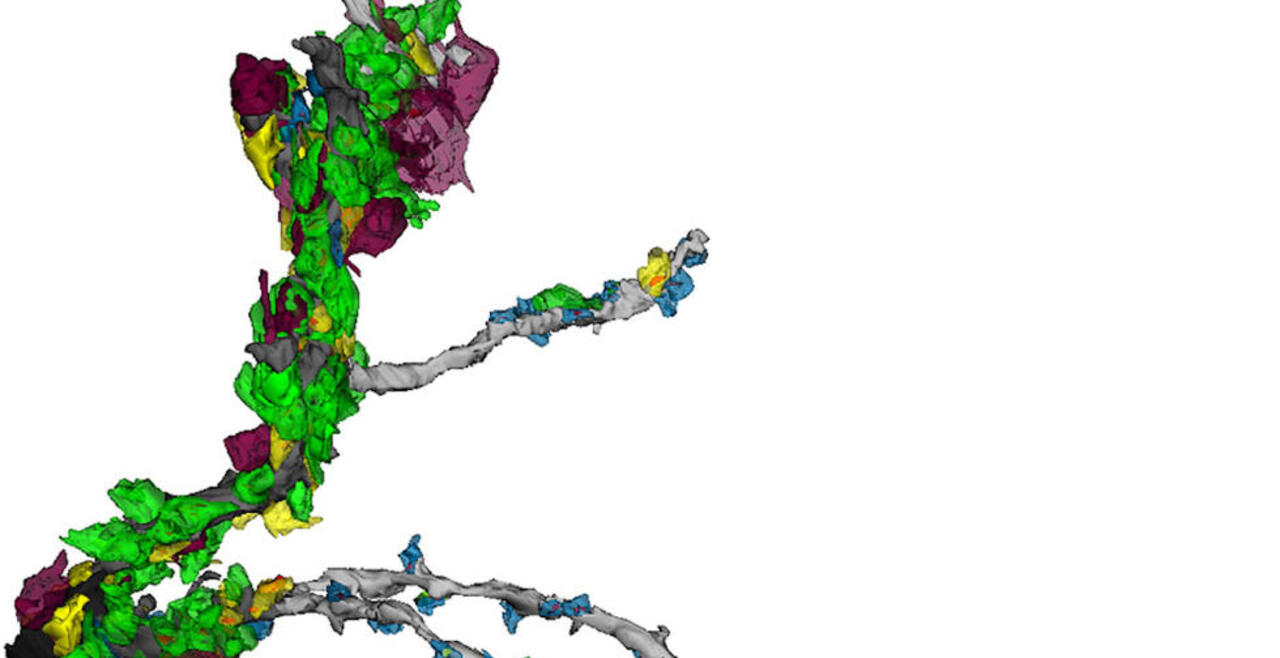
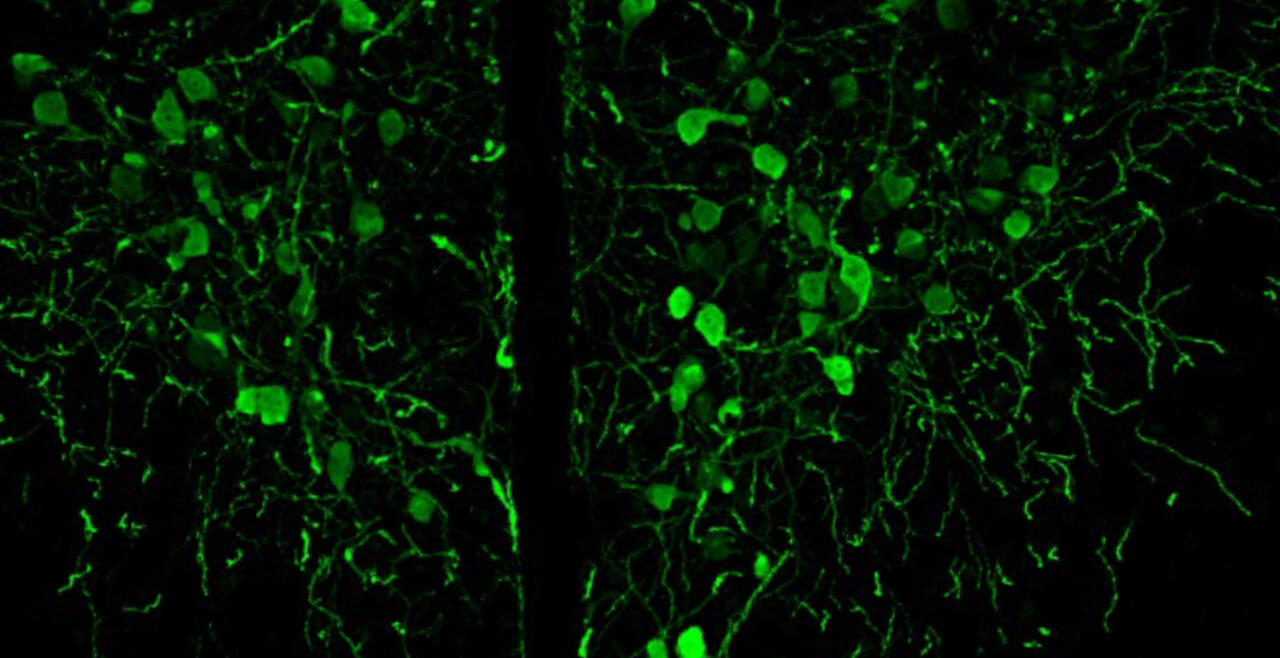
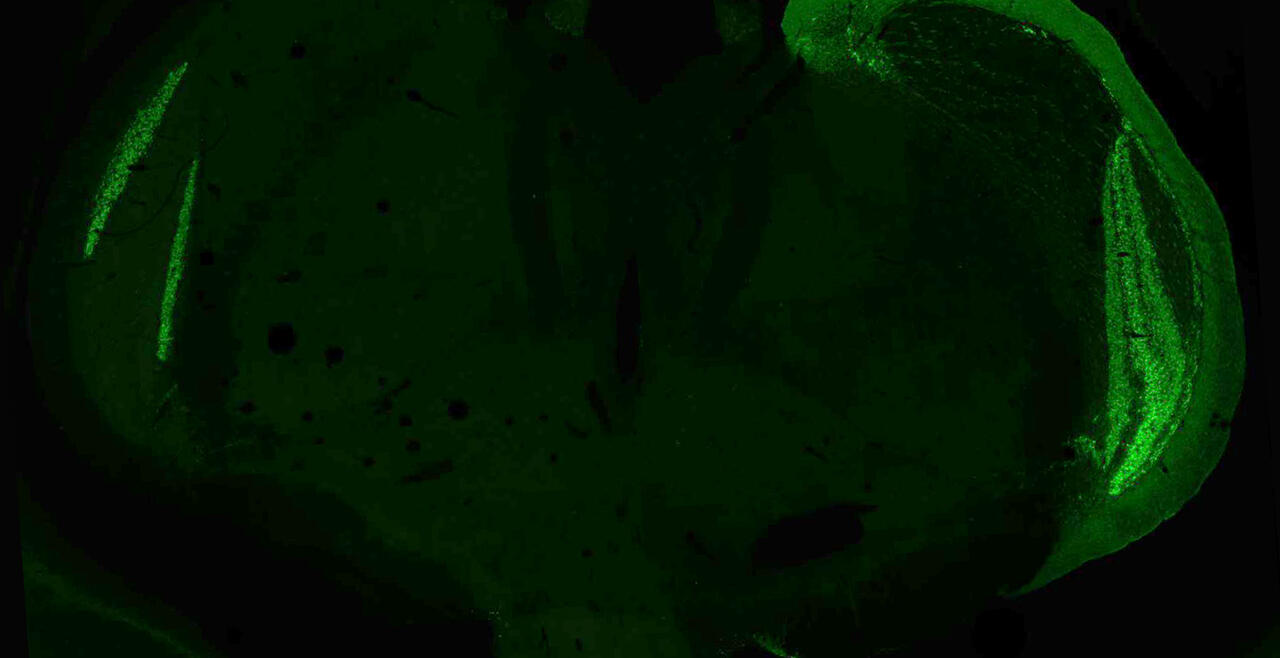
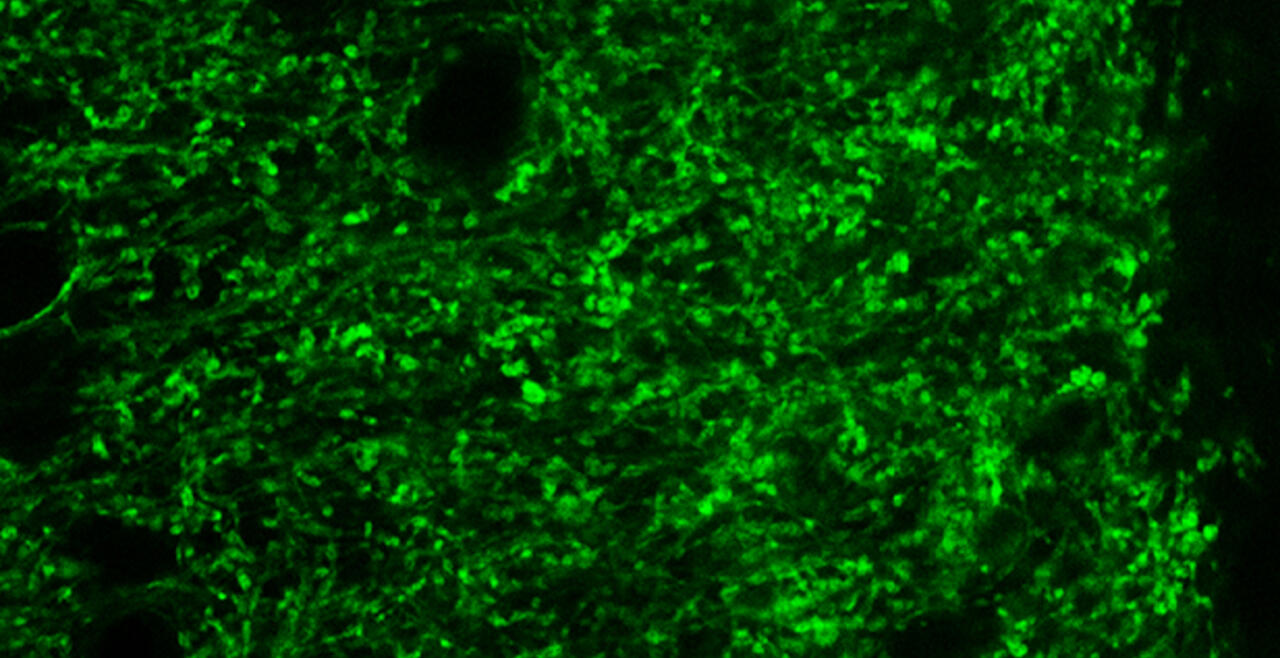
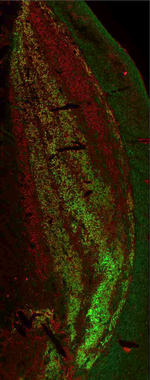
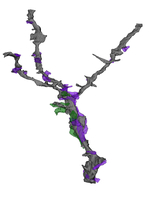
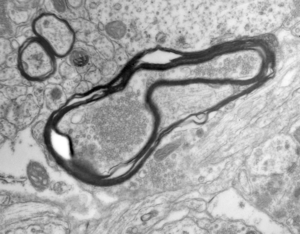
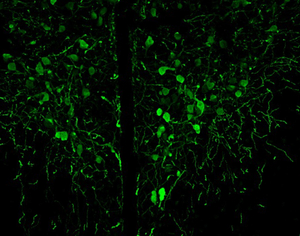
Synaptic connections among neurons that make up sensory pathways and other functional circuitries are the building blocks for how our brain functions, or in other words, how we see, hear, taste, move, learn, make plans or remember. These connections are amanable to change throughout life, allowing us to learn new skills, or to adapt to any external or internal alteration, including as we grow or age. By studying the morphological properties of neurons, synapses and identified axons, we aim to understand the synaptic inputs that converge on functionally distinct brain nuclei, how those inputs develop to form functional circuitries of the adult brain, and how they re-wire or degenerate.
What are the mechanisms by which the critical period of developmental plasticity is initiated, and terminated? Is there a change in the neurotransmitters, neuromodulators, hormones or their receptors that can signal the changes in sensory perception or the behavior? How do glial cells interact with neurons in developing or aging brains? Using anatomical techniques including immuno-electron microscopy, ultrastructural morphometry, tract-tracing and confocal microscopy, we study synaptic curcuitries in visual and gustatory sensory pathways during postnatal development, healthy adulthood and aging stages of our life span.