http://orcid.org/0000-0003-4747-9429
Google Scholar
45. Halford, G. C.; Sublett, A. M.; Personick, M. L. "Real-Time Electroanalytical Measurements of Dynamic Reaction Chemistry Provide Generalizable Mechanistic Considerations for Metal Nanoparticle Synthesis." J. Am. Chem. Soc. 2026, 148, 7280.
44. Halford, G. C.; Hertle, S.; N. Nambiar, H.; Personick, M. L. "Using Electrochemistry to Benchmark, Understand, and Develop Noble Metal Nanoparticle Syntheses." ACS Nanosci. Au 2025, 5, 240.
43. Personick, M. L. “Light as an Orthogonal Synthetic Parameter in Metal Nanoparticle Growth.” J. Phys. Chem. C. 2024, 128, 8131.
42. Halford, G. C.†; McDarby, S. P.†; Hertle, S.; Kiely, A. F.; Luu, J. T.; Wang, C. J.; Personick, M. L. “Troubleshooting the Influence of Trace Chemical Impurities on Nanoparticle Growth Kinetics via Electrochemical Measurements.” Nanoscale 2024, 16, 11038. † Authors contributed equally. (2024 Emerging Investigators Special Collection)
41. Personick, M. L.*; Jallow, A. A.; Halford, G. C.; Baker, L. A.* “Nanomaterials Synthesis Discovery via Parallel Electrochemical Deposition.” Chem. Mater. 2024, 36, 3034. (*co-corresponding authors)
40. Alba-Rubio, A. C.; Christopher, P.; Personick, M. L.; Stowers, K. J. “Recommendations to Standardize Reporting on the Synthesis of Heterogeneous Catalysts.” J. Catal. 2024, 429, 115259.
Independent Work at Wesleyan University (2015-2023)
39. Personick, M. L. “History and Fundamentals of the Colloidal Synthesis of Shaped Metal Nanoparticles.” In One Hundred Years of Colloid Symposia: Looking Back and Looking Forward, ACS Symposium Series, Vol. 1457, Nagarajan, R., Ed. American Chemical Society: Washington, 2023; pp 247-283.
38. Halford, G. C.; Personick, M. L. “Bridging Colloidal and Electrochemical Nanoparticle Growth with In Situ Electrochemical Measurements.” Acc. Chem. Res. 2023, 56, 1228-1238.
37. Argento, G. M.; Judd, D. R.; Etemad, L. L.; Bechard, M. M.; Personick, M. L. “Plasmon-Mediated Reconfiguration of Twin Defect Structures in Silver Nanoparticles.” J. Phys. Chem. C. 2023, 127, 3890-3897.

36. Wang, C. J.; Shapiro, E. F.; Personick, M. L. “Halide Ions on Metal Nanoparticles for Shape- and Composition-Controlled Synthesis.” In Encyclopedia of Nanomaterials (1st Edition), Yin, Y.; Lu, Y.; Xia, Y., Eds. Elsevier: Oxford, 2023; pp 263-276.

35. Personick, M. L., “Plasmon-Mediated Syntheses of Polyhedral Noble Metal Nanoparticles.” In World Scientific Reference on Plasmonic Nanomaterials, Wang, J., Ed. World Scientific: Singapore, 2022; Vol. 22, pp 47-88.
34. McDarby, S. P.; Personick, M. L. “Potential-Controlled (R)Evolution: Electrochemical Synthesis of Nanoparticles with Well-Defined Shapes.” ChemNanoMat 2022, 8, e202100472.
33. McDarby, S. P.; Wang, C. J.; King, M. E.; Personick, M. L. “An Integrated Electrochemistry Approach to the Design and Synthesis of Polyhedral Noble Metal Nanoparticles.” J. Am. Chem. Soc. 2020, 142, 21322-21335. Supplementary cover article.

32. Habib, A.†; King, M. E.†; Etemad, L. L.; Distler, M. E.; Morrissey, K. H.; Personick, M. L. “Plasmon-Mediated Synthesis of Hybrid Silver-Platinum Nanostructures.” J. Phys. Chem. C 2020, 124, 6853-6860. †Authors contributed equally.
31. King, M. E.; Kent, I. A.; Personick, M. L. “Halide-Assisted Metal Ion Reduction: Emergent Effects of Dilute Chloride, Bromide, and Iodide in Nanoparticle Synthesis.” Nanoscale 2019, 11, 15612-15621.
30. Jung, H.†; King, M. E.†; Personick, M. L. “Strategic Synergy: Advances in the Shape Control of Bimetallic Nanoparticles with Dilute Alloyed Surfaces.” Curr. Opin. Colloid Interface Sci. 2019, 40, 104-117. † Authors contributed equally.
29. Robertson, D. D.; Personick, M. L. “Growing Nanoscale Model Surfaces to Enable Correlation of Catalytic Behavior Across Dissimilar Reaction Environments.” Chem. Mater. 2019, 31, 1121-1141. ("Up and Coming" Perspective Series) Front cover article.

28. King, M. E; Personick, M. L. “Iodide-Induced Differential Control of Metal Ion Reduction Rates: Synthesis of Terraced Palladium-Copper Nanoparticles with Dilute Bimetallic Surfaces.” J. Mater. Chem. A 2018, 6, 22179-22188. (2018 Emerging Investigators Issue)
27. Stone, A. L.; King, M. E.; McDarby, S. P.; Robertson, D. D.; Personick, M. L. “Synthetic Routes to Shaped AuPt Core-Shell Particles with Smooth Surfaces Based on Design Rules for Au Nanoparticle Growth.” Part. Part. Syst. Charact. 2018, 35, 1700401.
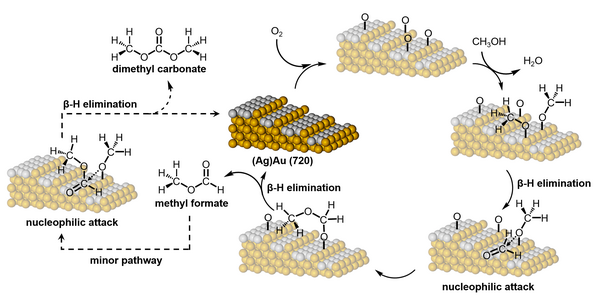
26. Robertson, D. D.; King, M. E.; Personick, M. L. “Concave Cubes as Experimental Models of Catalytic Active Sites for the Oxygen-Assisted Coupling of Alcohols by Dilute (Ag)Au Alloys.” Top. Catal. 2018, 61, 348-356.
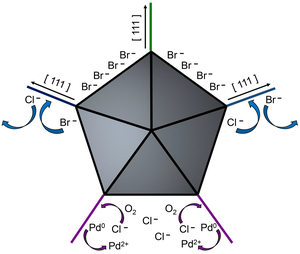
25. King, M. E.; Personick, M. L. “Defects by Design: Synthesis of Palladium Nanoparticles with Extended Twin Defects and Corrugated Surfaces.” Nanoscale 2017, 9, 17914-17921.
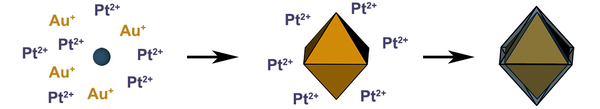
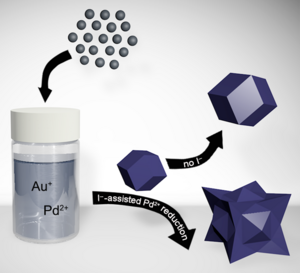
24. King, M. E.; Personick, M. L. “Bimetallic Nanoparticles with Exotic Facet Structures via Iodide-Assisted Reduction of Palladium.” Part. Part. Syst. Charact. 2017, 34, 1600422. Inside cover article.
Publications from Postdoctoral, Graduate, and Undergraduate Work
23. Personick, M. L.; Madix, R. J.; Friend, C. M. “Selective Oxygen-Assisted Reactions of Alcohols and Amines Catalyzed by Metallic Gold: Paradigms for the Design of Catalytic Processes.” ACS Catal. 2017, 7, 965. Cover article.
22. Personick, M. L.; Montemore, M. M.; Kaxiras, E.; Madix, R. J.; Biener, J.; Friend, C. M. “Catalyst Design for Enhanced Sustainability Through Fundamental Surface Chemistry.” Phil. Trans. R. Soc. A 2016, 374, 20150077. Cover article.
21. Wang, L.-C.; Personick, M. L.; Karakalos, S.; Fushimi, R.; Friend, C. M.; Madix, R. J. “Active Sites for Methanol Partial Oxidation on Nanoporous Gold Catalysts.” J. Catal. 2016, 344, 778.
20. Padmos, J. D.; Personick, M. L.; Tang, Q.; Duchesne, P. N.; Jiang, D.; Mirkin, C. A.; Zhang, P. “The Surface Structure of Silver-coated Gold Nanocrystals and Its Influence on Shape Control.” Nat. Commun. 2015, 6, 7664.
19. Wang, L.-C.; Stowers, K. J.; Zugic, B.; Personick, M. L.; Biener, M. M.; Biener, J.; Friend, C. M.; Madix, R. J. “Exploiting Basic Principles to Control the Selectivity of the Vapor Phase Catalytic Oxidative Cross-coupling of Primary Alcohols over Nanoporous Gold Catalysts.” J. Catal. 2015, 329, 78.
18. Personick, M. L.; Zugic, B.; Biener, M. M.; Biener, J.; Madix, R. J.; Friend, C. M. “Ozone-Activated Nanoporous Gold: A Stable and Storable Material for Catalytic Oxidation.” ACS Catal. 2015, 5, 4237.
17. Personick, M. L.; Mirkin, C. A. “Making Sense of the Mayhem Behind Shape Control in the Synthesis of Gold Nanoparticles.” J. Am. Chem. Soc. 2013, 135, 18238.
16. Young, K. L.; Personick, M. L.; Engel, M.; Damasceno, P. F.; Barnaby, S. N.; Bleher, R.; Li, T.; Glotzer, S. C.; Lee, B.; Mirkin, C. A. “A Directional Entropic Force Approach to Assemble Anisotropic Nanoparticles into Superlattices.” Angew. Chem., Int. Ed. 2013, 52, 13980.
15. Liu, G.; Young, K. L.; Liao, X.; Personick, M. L.; Mirkin, C. A. “Anisotropic Nanoparticles as Shape-Directing Catalysts for the Chemical Etching of Silicon.” J. Am. Chem. Soc. 2013, 135, 12196.
14. Langille, M. R.; Personick, M. L.; Mirkin, C. A. “Plasmon-Mediated Syntheses of Metallic Nanostructures.” Angew. Chem., Int. Ed. 2013, 52, 13910.
13. Shin, Y. J.; Ringe, E.; Personick, M. L.; Cardinal, M. F.; Mirkin, C. A.; Marks, L. D.; Van Duyne, R. P.; Hersam, M. C. “Centrifugal Shape Sorting and Optical Response of Polyhedral Gold Nanoparticles.” Adv. Mater. 2013, 25, 4023.
12. Personick, M. L.; Langille, M. R.; Wu, J.; Mirkin, C. A. “Synthesis of Gold Hexagonal Bipyramids Directed by Planar-Twinned Silver Triangular Nanoprisms.” J. Am. Chem. Soc. 2013, 135, 3800.
11. Personick, M. L.; Langille, M. R.; Zhang, J.; Wu, J.; Li, S.; Mirkin, C. A. “Plasmon-Mediated Synthesis of Silver Cubes with Unusual Twinning Structures Using Short Wavelength Excitation.” Small 2013, 9, 1947.
10. Rycenga, M.; Langille, M. R.; Personick, M. L.; Ozel, T.; Mirkin, C. A. “Chemically Isolating Hotspots on Concave Nanocubes.” Nano Lett. 2012, 12, 6218.
9. Langille, M. R.†; Personick, M. L.†; Zhang, J.; Mirkin, C. A. “Defining Rules for the Shape Evolution of Gold Nanoparticles.” J. Am. Chem. Soc. 2012, 134, 14542. † Authors contributed equally.
8. Langille, M. R.; Zhang J.; Personick, M. L.; Li, S.; Mirkin, C. A. “Stepwise Evolution of Spherical Seeds into 20-Fold Twinned Icosahedra.” Science 2012, 337, 954.
7. Personick, M. L.; Langille, M. R.; Zhang, J.; Mirkin, C. A. “Shape Control of Gold Nanoparticles by Silver Underpotential Deposition.” Nano Lett. 2011, 11, 3394.
6. Langille, M. R.; Personick, M. L.; Zhang, J.; Mirkin, C. A. “Bottom-Up Synthesis of Gold Octahedra with Tailorable Hollow Features.” J. Am. Chem. Soc. 2011, 133, 10414.
5. Personick, M. L.; Langille, M. R.; Zhang, J.; Harris, N.; Schatz, G. C.; Mirkin, C. A. “Synthesis and Isolation of {110}-Faceted Gold Bipyramids and Rhombic Dodecahedra.” J. Am. Chem. Soc. 2011, 133, 6170.
4. Zhang, J.†; Langille, M. R.†; Personick, M. L.; Zhang, K.; Li, S.; Mirkin, C. A. “Concave Cubic Gold Nanocrystals with High-Index Facets.” J. Am. Chem. Soc. 2010, 132, 14012.
3. Choi, S.; Personick, M. L.; Bogart, J. A.; Ryu, D.; Redman, R. M.; Laryea-Walker, E. “Oxidation of a Guanine Derivative Coordinated to a Pt(IV) Complex Initiated by Intermolecular Nucleophilic Attacks.” Dalton Trans. 2011, 40, 2888.
2. Pirzada, Z.; Personick, M.; Biba, M.; Gong, X.; Zhou, L.; Schafer, W.; Roussel, C.; Welch, C. J. “Systematic Evaluation of New Chiral Stationary Phases for Supercritical Fluid Chromatography using a Standard Racemate Library.” J. Chromatogr. A 2010, 1217, 1134.
1. Choi, S.; Vastag, L.; Larrabee, Y.C.; Personick, M. L.; Schaberg, K. B.; Fowler, B. J.; Sandwick, R. K.; Rawji, G. “The Importance of Pt(II) Catalyzed Pt(IV) Substitution for the Oxidation of Guanosine Derivatives by Pt(IV) Complexes.” Inorg. Chem. 2008, 47, 1352

