Research
The ultimate goal of our laboratory is to improve human health through the development of a better understanding of the cellular processes that lead to disease and to leverage this understanding to improve disease diagnosis and treatment. These multidisciplinary efforts are supported by a combination of approaches including synthesis, protein design and evolution, bioanalytical chemistry, molecular and cell biology, and animal studies.
Near-Infrared Dyes
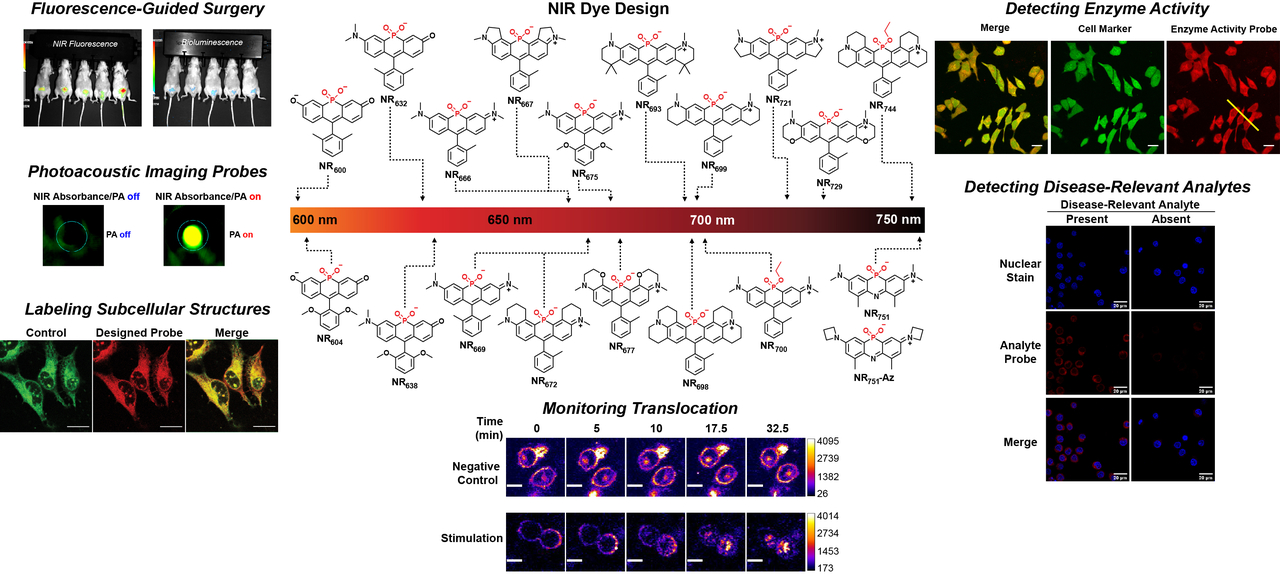
Near-infrared (NIR) light can penetrate into living organisms and dyes that absorb within this wavelength range can provide spatially resolved, real-time information. Our laboratory is interested in expanding the palette and chemical diversity of NIR dyes to improve their properties. Towards this goal, we have developed new NIR dyes that display remarkable brightness and photostability. These scaffolds are being used to develop reagents for fluorescence-guided surgery, deep tissue imaging, and molecular imaging.
Targeted Drug Delivery
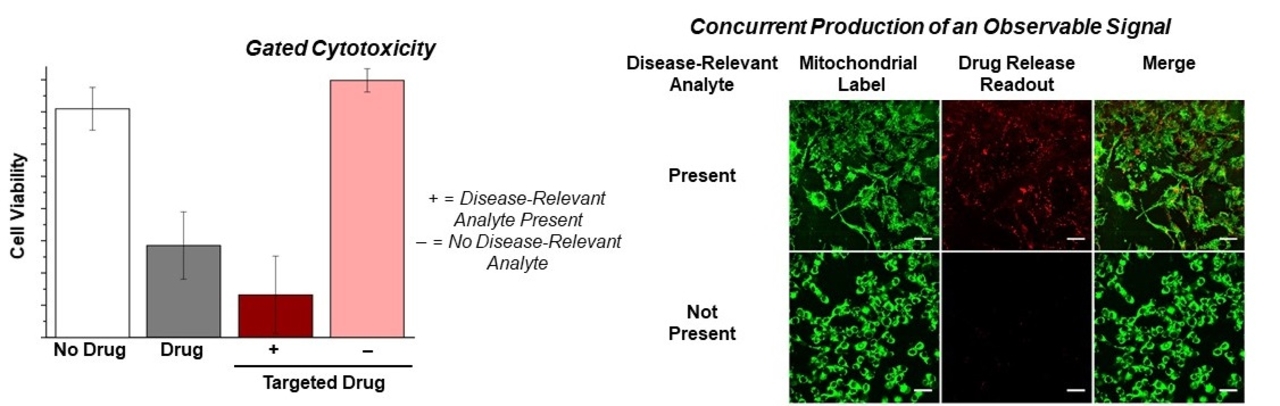
Disease processes result in altered cellular biochemistry. Often, new analytes are produced in diseased cells and provide a potential marker for targeting cytotoxic compounds. Using activity-based sensing approaches, we are engineering functional molecules that can sense the altered biochemistry of diseased cells and release cytotoxic compounds. Concurrent production of a NIR dye provides the ability to assess the amount of compound delivered to target cells and visualization of potential off-target sites.
Dissecting Cellular Communication
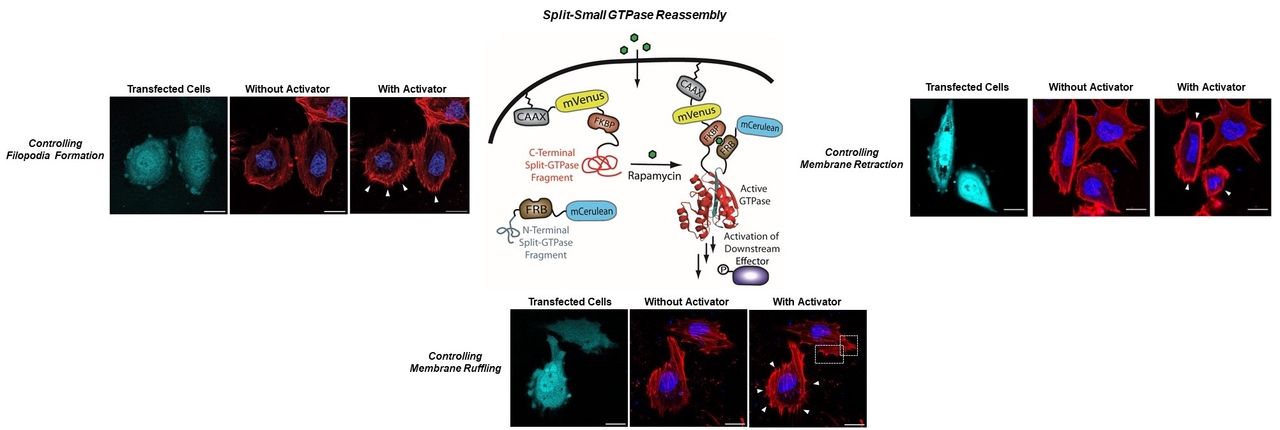
Split-Small GTPases: The development of standardized parts for controlling signaling pathways could allow new functions to be engineered into living systems as well as enable fundamental studies of cellular signaling pathways. To realize these goals, new approaches to controlling protein function are needed. Our laboratory is utilizing split-protein reassembly to develop a lexicon of split-small GTPases to control a diverse array of cellular functions. These tools are being used to dissect signaling pathways involved in human disease.
Cellular Protein Tyrosine Phosphatase Probes: Protein tyrosine phosphatases (PTPs) catalyze the removal of phosphate groups from tyrosine. Importantly, members of this enzyme family are now established as nodes in human disease, making PTPs attractive targets for the development of diagnostics as well as inhibitors. However, the role of many PTPs in cellular signaling remains poorly understood due to the lack of approaches to monitor the activity of these enzymes in living cells. Our laboratory is addressing the need for tools in this area by developing approaches to assess PTP activity within living cells. These new tools will provide insight into PTP activity dynamics and spatially restricted signaling. In addition, these probes can be used to screen for PTP inhibitors that are active in living cells.